Salt Safety Inspection
Detect chalk powder and white stone powder in common salt
Inspection Guide
Click to enlarge
Salt Purity Detection
Common salt is sometimes adulterated with chalk powder or finely ground white stone powder to increase weight and improve whiteness.
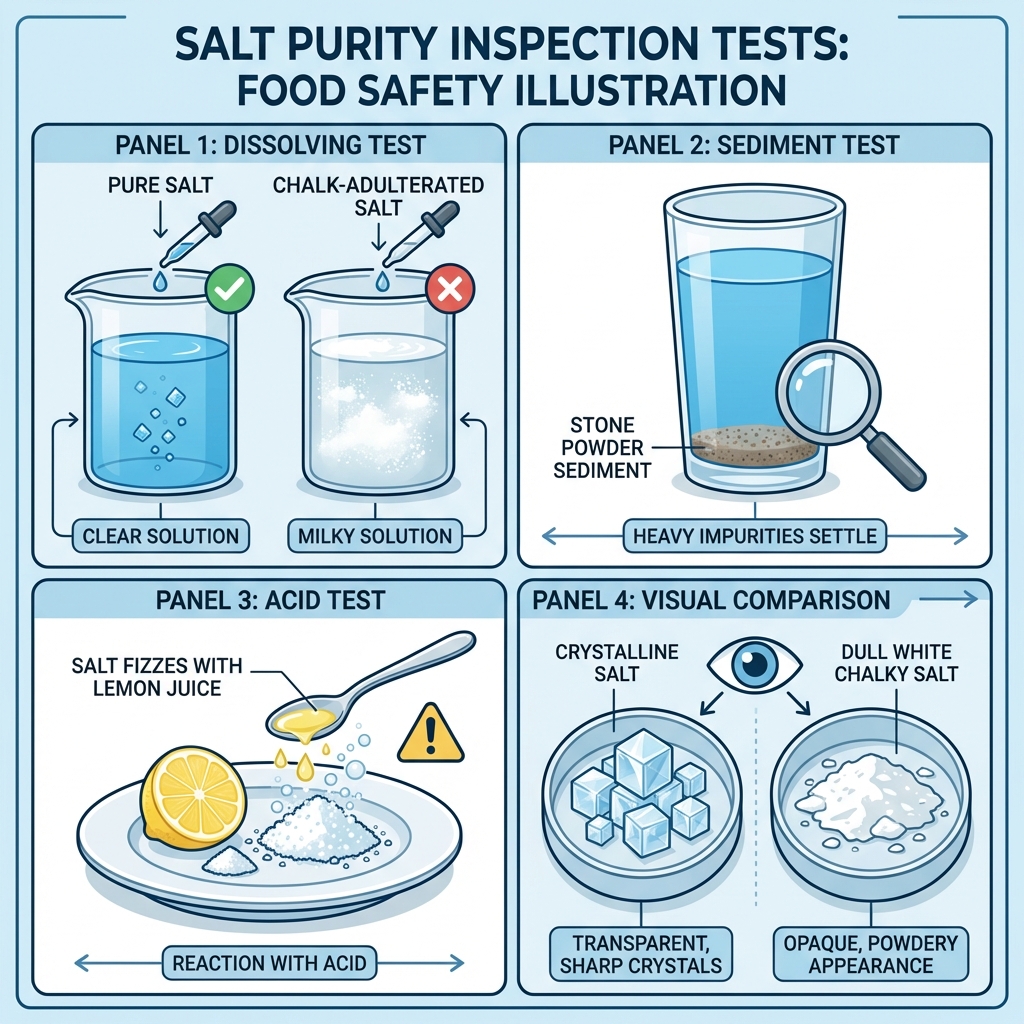
1. The Dissolving Test (Water Test):
Stir a spoonful of salt in a glass of clear water. Pure salt will dissolve completely, leaving the water clear. If the water turns milky white, it indicates the presence of chalk powder. If particles settle at the bottom, it's white stone powder or sand.
2. The Acid Test (HCl/Vinegar):
Take a small amount of salt and add a few drops of lemon juice or vinegar (mild acid). If the salt starts to fizz or produces bubbles of gas, it confirms the presence of chalk powder (calcium carbonate).
3. The Texture and Grittiness:
Rub a small amount of salt between your dry fingers. Pure table salt should feel uniform. If you feel very hard, gritty, or sharp particles that don't break down, it's likely stone dust or sand.
4. Visual Appearance:
Pure salt crystals have a characteristic crystalline shine. Chalk powder makes the salt look dull and very white, lacking the crystalline structure.
Quick Safety Tips
- Salt should dissolve completely in water
- Milky water indicates chalk powder adulteration
- Check for undissolved sediment at the bottom
- Avoid salt that fizzes with lemon juice
Chemical Concerns
Step 1: AI Visual Scan
New to BioLens? Try a sample test
